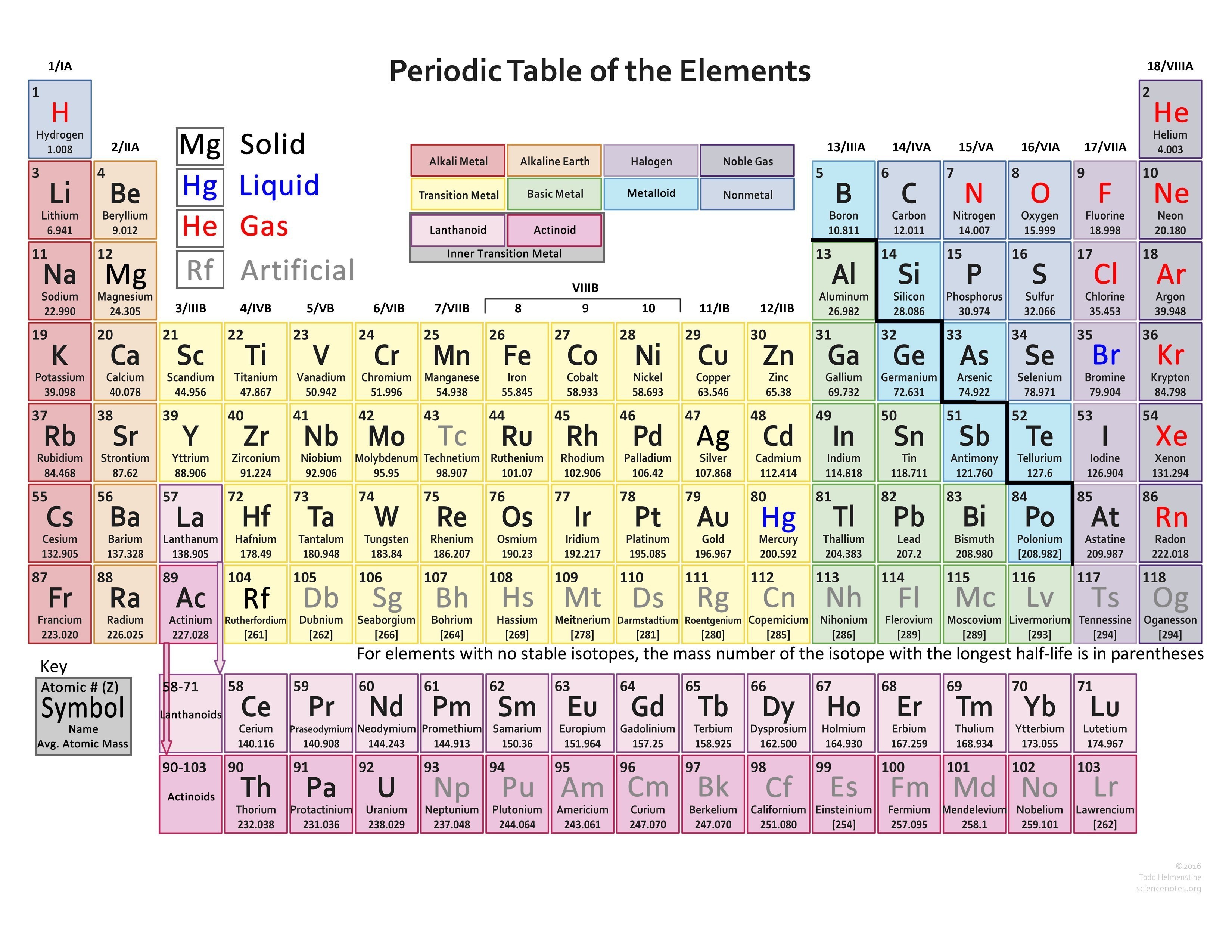
The blocksĮxplain the electronic configuration of the elements as well as their chemical properties. Elements within these blocks areĬategorised in accordance with the orbital occupied by the outermost electron. The periodic table is split into 4 blocks S,P,D and F. Organisation in the table, and allows understanding of chemical reactions in elements.įigure 1: Periodic Table (Free Printable periodic tables (pdf and PNG) 2023)Įxplain the significance of the S, P and D blocks in the periodic table. The element properties, the groups are named, allowing there to be categorising and The overall significance of the main groups and number of the periods is the predictability of Less reactive as it is harder to remove 18 electrons than 1. Highly reactive, it has 1 electron in the outer shell, the noble gases having 18 electrons makes it 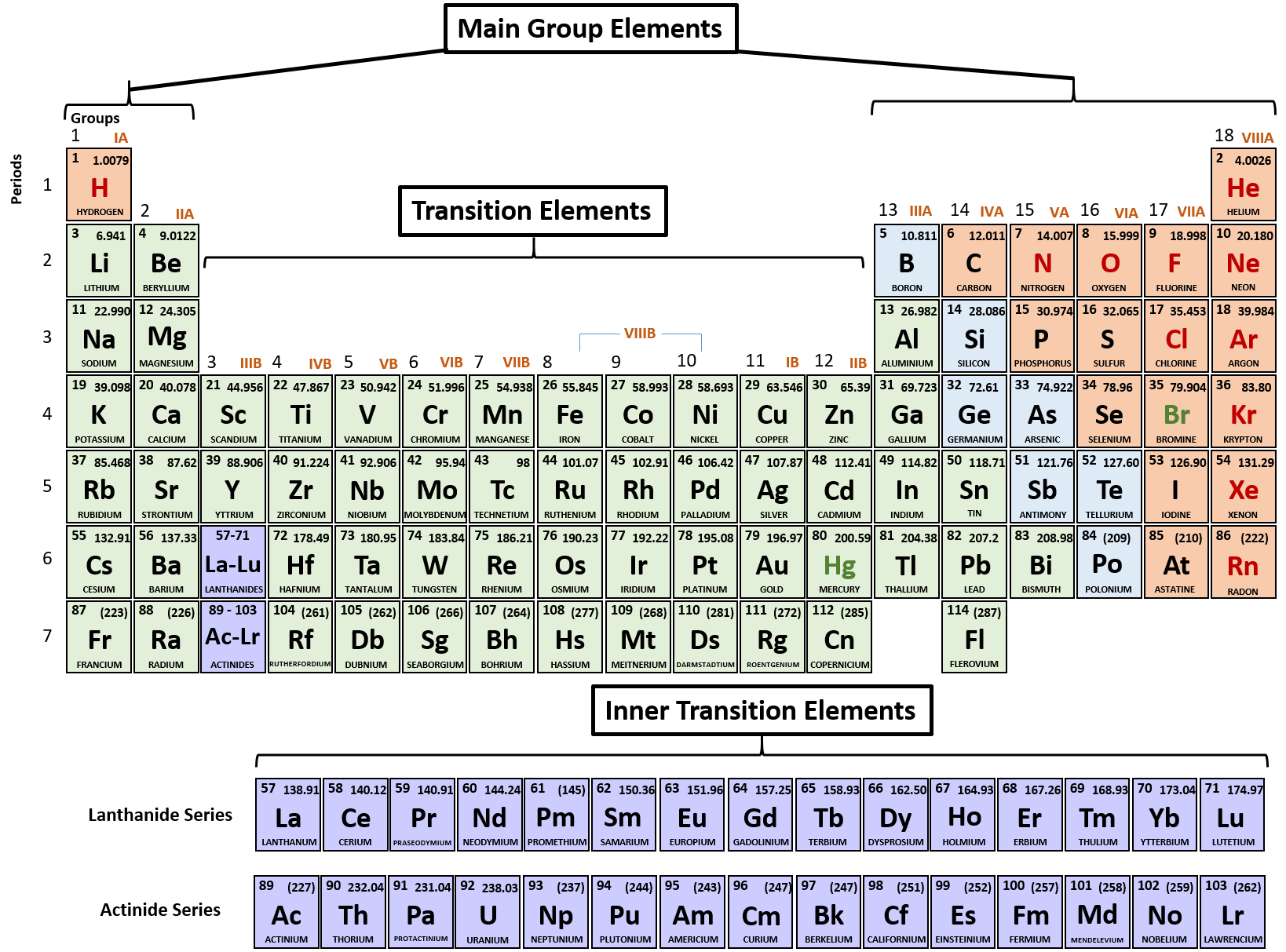
The group begin with group 1 being alkali metals and ending with noble gases at 18. The ability to readily loose theĮlectrons on the shell contribute to the chemical reactions. The number of shells occupied determineĮach elements atomic radius, its reactivity and ionisation energy. In the outermost shell shows which group it is in. The period number is the number of shells around the nucleus, the number of electrons The periodic table gives an atoms electron configuration based upon its location within the The groups run in vertical columns from 1 to 18.Įlements are put in order of their atomic number, meaning elements with similar propertiesĪppear in the same vertical column or group, properties such as melting point, melting point,ĭensity, and hardness. Horizontal rows and are numbered 1 down to 7. Predicted in the elements thus predicting reactions between each element. The position of the elements within the table enables properties to be predicted. Officially, the valence electrons are the electrons in the outer shell ofĮlectrons in its outer shell and so can represent it as a “Cl” with sevenįirst shell and the 8 electrons in the second are inner shell electrons and areĬhart of the main group elements and their Lewis dot symbols.Bethan White | Unit 8 | 15/11/ Contents PageĮxplain the significance of the main groups and numbering of the periods. The outer shell are given the name valence electrons. Represent the number of electrons in the outer shell with dots. The third shell can only hold a maximum of 18 electrons but isīecause it is the outer shells that react, we are most interested in the The second shell can only hold a maximum of 8 electrons. The first shell is the smallest so it can only hold a maximum of 2 electrons. Shells correspond to the orbits of the Bohr model. The first two quantum numbers n (the shell) and l (the subshell) are both important in It is the outer shell electrons that form these bonds betweenĪtoms. There are some elements that show some, but not all, of the metallicĬalled metalloids and are labeled here are semi-conductors.Įlectrons are the “glue” that hold atoms together in compounds. Nonmetals typically do not show these properties. Have a metallic sheen (shiny) are malleable (bendable) and conduct electricity. The elements can also be divided into two main groups, the metals and the The columns with A (IA through VIIIA) are called the main group With B (IB through VIIIB) are called the transition elements. The metals in group IB (copper, silver and gold) are sometimes called the The elements in group VIIA are called the halogens and theĮlements in group VIIIA are called the noble gases or the inert gases. 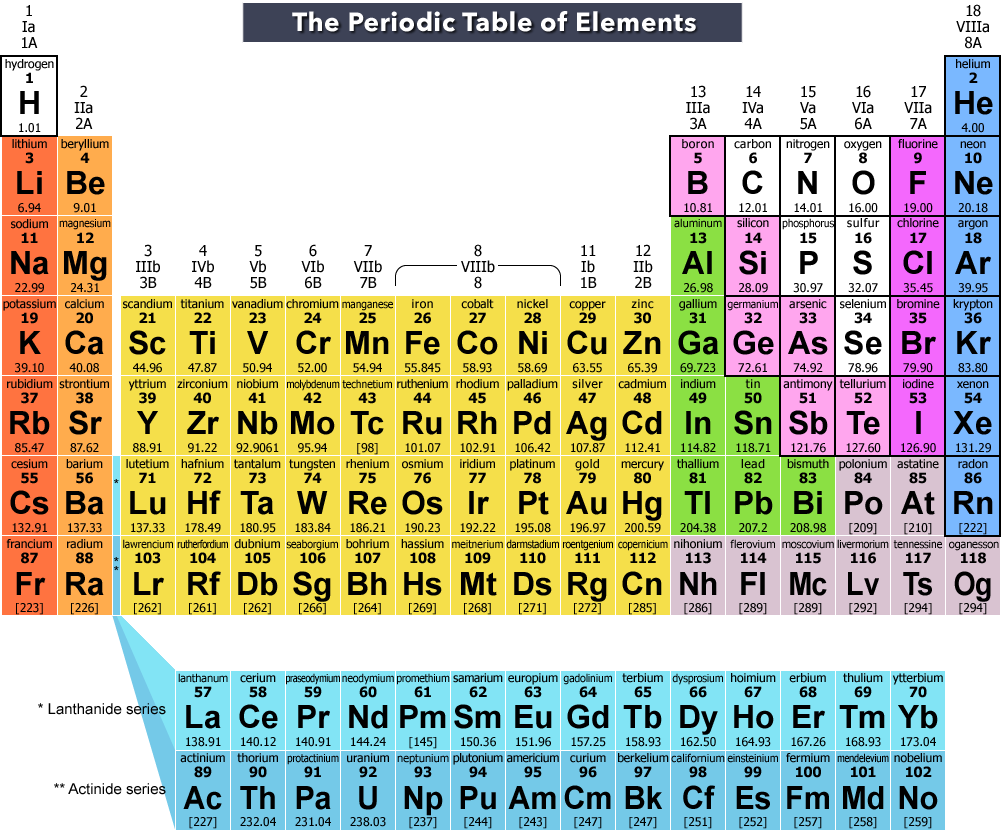
The elements in group IIA are called the alkaline earth metals. The elements in group IA are called the alkali metals. Elements with similar reactivity are put into the same column or group. Elements are arranged by reactivity in the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
